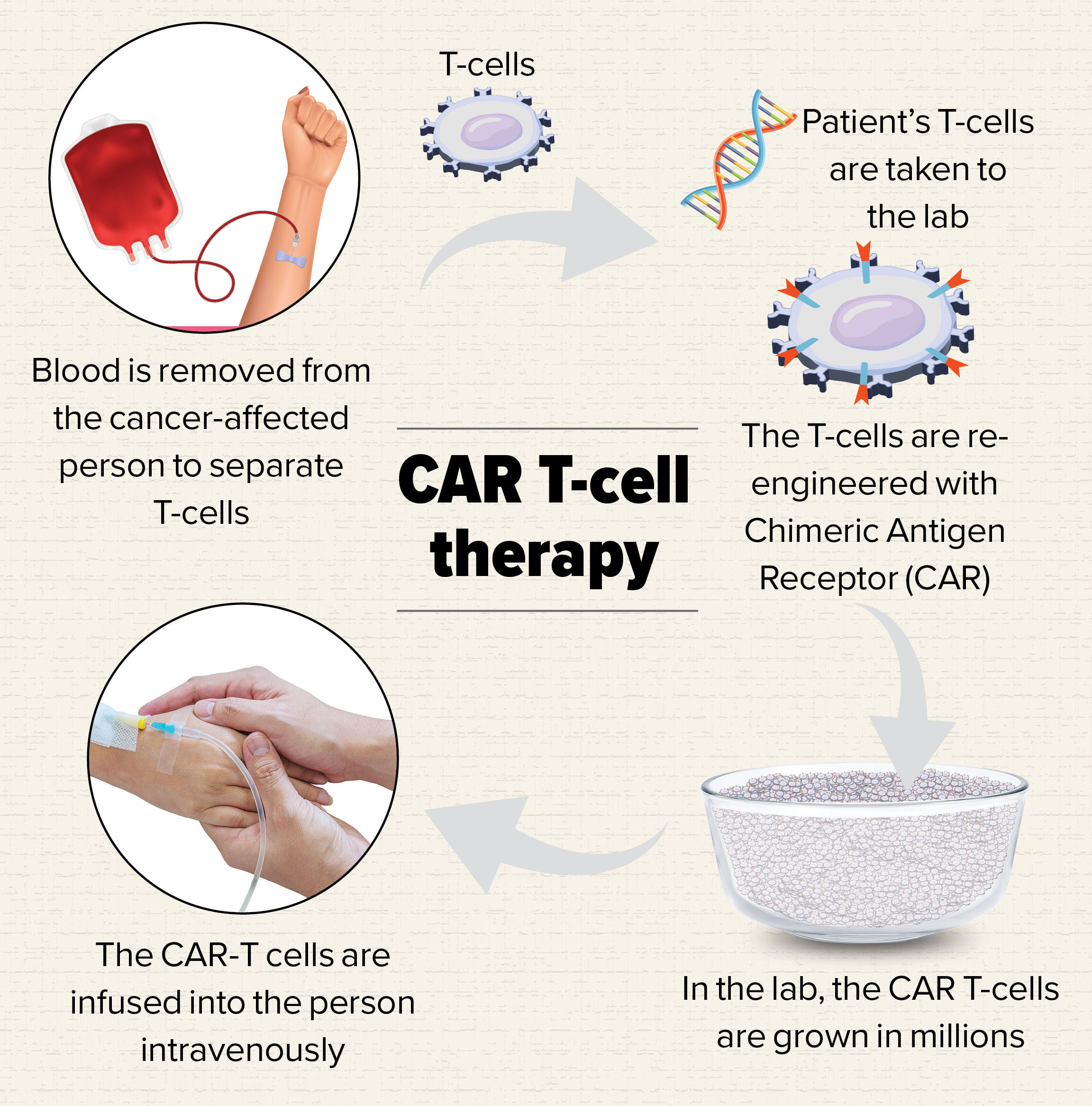
Imagine making cancer cells disappear with an intravenous insertion of the affected person’s own re-engineered blood cells. That’s exactly what researchers from the Indian Institute of Technology Bombay and the Tata Memorial Centre, Mumbai, have achieved — indigenously and at a lower cost. CAR T-cell therapy — or chimeric antigen receptor T-cell therapy — is a type of gene therapy in which an affected person’s own immune cells are re-engineered to fight with cancer cells.
Collaborative work between the two has led to the production of a CAR T-cell product that has been found to be safe and has shown promising early signs of efficacy in a recently concluded Phase 1 trial.
The Mumbai research has, for the first time, come up with a made-in-India CAR T-cell therapy for blood cancers such as lymphoma and leukemia. While a drug is available in the US, the Indian researchers worked on creating an indigenous product that has fewer side effects and is cost-effective.
The project started in 2014, and while the product development took place at IIT Bombay, the clinical trial took place at the Tata Memorial Centre. Dr (surg cdr) Gaurav Narula, principal investigator of paediatric acute lymphocytic leukaemia, and Dr Hasmukh Jain, principal investigator of adult B-cell lymphoma, both from Tata Memorial Centre, recruited patients for the trials.
Rahul Purwar, associate professor, IIT Bombay, told Happiest Health that the indigenous drug has been found to be safe in Phase 1 of the trial. “CAR T-cells, which are also called ‘living drugs’, are personalised drugs in the sense that we take the patient’s immune cells and genetically modify them in the laboratory. Then we expand the cells, and infuse them back into the patient,” said Dr Purwar, who spearheaded the project along with Dr Narula. Dr Purwar is also founder of ImmunoACT, an advanced cell and gene therapy company.
The researchers are now gathering data for the publication of the research findings of the Phase 1 trial in a scientific journal. “Whenever any new medical product has to come to the market, first we have to do a Phase 1 clinical trial to check the safety of the drug, which we’ve achieved now,” said Dr Purwar.
Explaining the results of the Phase 1 trial at a recent medical conference of the Asia Pacific Blood and Bone Marrow Transplantation Group in Kochi, Kerala, Dr Narula said the CAR T-cell product involved animal studies and the clinical grade product development was done by the researchers collaboratively.
“The challenge of initiating the CAR T-cell therapy helped create a roadmap for cell therapies in India,” Dr Narula at the conference.
Dr Narula told Happiest Health that they recruited those patients for the study who did not respond to the initial cancer treatment, had faced relapse and were ineligible for any further curative therapy.
A total of 16 patients with cancer were chosen for Phase 1 of the research — ten of them had lymphoma and six, leukemia. While all the lymphoma patients were adults (above 18 years), the leukemia subjects were between the ages of three and 25.
Half of the leukaemia patients who were treated were found to have negative results in the test for minimal residual disease (MRD). A negative MRD means no cancer cells were detected. The test is also a measure for relapse. Two more leukaemia patients had significant reduction in cancer burden, close to MRD levels.
Three of the six leukemia patients — a girl and a boy aged eight, and a boy aged 16 — had complete response, meaning their bone marrow was found to be cancer-free one month after the treatment.
“Half the leukemia patients who were part of the Phase 1 trial became cancer-free,” said Dr Narula. There was no response in the case of one leukemia patient.
“The overall response rate we have achieved among the leukemia patients was 86 per cent,” said Dr Narula.
The response time in CAR-T cell treatment is one month, said Dr Purwar. That is, those treated with CAR T-cells are followed for one month to check the response. “In those with leukaemia, the MRD tests are done a month after the IV infusion of the CAR T-cells,” said Dr Purwar. “Reduction in tumour burden is seen in lymphoma patients through the CT scan done after 30 days of the treatment.”
However, asked how the CAR T-cell therapy would prevent remission, the researchers said they currently had data of one year and it needed to be watched further.
CAR T-cell therapy: ‘One-time treatment’
Dr Purwar said CAR T-cell therapy is a one-time treatment — unlike chemotherapy and immunotherapy, which are given for a long time. “It’s given through IV [drip],” he said. “The quantity of the CAR T-cells introduced varies – it’s in a range of one million to five million CAR T-cells given per kg weight of the patient.
“Treatment is given only once and they [the CAR T-cells] stay for long time and that’s the reason they’re called ‘live’ drugs,” said Purwar. “IIT Bombay is working on the product design and product validation. The Tata Memorial Centre is doing the clinical trials. So far, we have just concluded the Phase 1 for two indications: leukemia for kids and lymphoma for adults.
“We are awaiting approval from the government of India, the Central Drugs Standard Control Organisation, to begin Phase 2 of the clinical trial.”
Dr Narula said the primary objective had been met in Phase 1 by achieving safety. “Now, in Phase 2, the objective will be to achieve efficacy along with observing safety in a large number of subjects — maybe more than 50 cancer patients,” said Dr Narula.
Reaching the market
Patented as HRC9-19, the made-in-India CAR T-cell therapy drug is likely to be available in the market by early 2024.
“We think that the product would cost 1/10th or 1/15th of the cost of available treatment in the US,” said Dr Purwar. “In the US, it costs half a million dollars (nearly Rs 4 crore). We are thinking of keeping it at $25,000 to $30,000, which roughly translates to Rs 25 lakh to Rs 30 lakh.”
Aware that affordability would still be a factor, the researchers hope the cost would come down in the future with technological advancement. “There will be better pricing within a few years, when the technology further matures,” said Dr Purwar.

















2 Responses
Can children suffering from blood cancer, asked for go for bone marrow transplant be a part of the clinical trial?
Thank you for your query, we at Happiest Health however do not offer medical advice or suggest any doctors’ names.