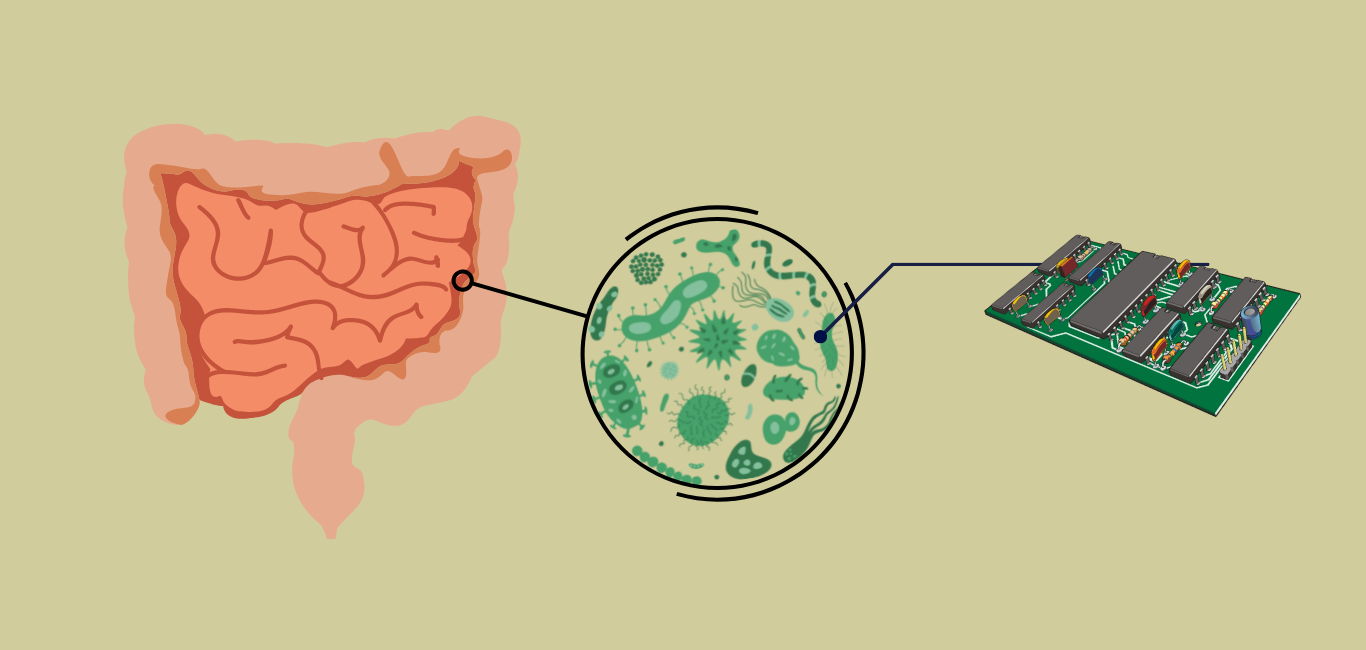
Think of the gut like a forest filled with trees, birds and insects all living off each other symbiotically. Like this environment, the gut is home to millions of species of microbes each interacting closely with the cells in the gut. These interactions serve important intestinal functions, helping to digest food, absorb important nutrients and drugs and offering protection against infectious agents.
Scientists are yet to understand the depth of functions that the human gut plays in health and disease. Today, they hope to solve this by recreating the gut on a chip – to mimic the way our gut (and its inhabitants) function and how different environments, medications, food, stress and so on affect it. This organ-on-chip model will help researchers/clinicians develop better therapeutics and study diseases that affect the gut.
“By collecting and culturing a person’s own cells, we can create an individual-specific gut environment using the gut-on-a-chip model. Every individual has a different genetic background, different reactions against different therapies, and different adverse effects. It will be extremely useful if we could predict the therapeutic efficacy and safety using your own cells” says Dr Woojung Shin, post-doctoral researcher at the Wyss Institute of Harvard. She has been working on developing accurate organ-on-chip models to study how organs function.
The issues with current studies of organs
Using a microfluidic chip to recapture how the gut behaves in the body has been a new venture spearheaded mainly by the failure of current model systems such as animals and 2D culture systems used in preclinical studies in clinical trials.
“Scientists have realized that the responses that animals give to a drug do not really mimic the human responses. So that is associated with a very high degree of drug failure in the clinical trials before they’re able to reach the market,” says Dr Prajakta Dandekar Jain, assistant professor, Department of Pharmaceutical Sciences and Technology, at the Institute of Chemical Technology (ICT), Mumbai. Her lab works with microfluidic systems to develop skin-on-chip models.
It is estimated that 90% of clinical drug development fails and the cost of developing a new compound is 1-2 billion dollars, should it pass through all phases of the clinical trials.
But with the use of microfluidic devices, researchers have been able to make miniature versions of organs on a chip.
The chip is made of transparent silicone material, not bigger than a USB memory stick. It contains tiny hollow channels lined with human cells taken from organs. These minuscule channels allow the flow of nutrients and oxygen through them allowing us to imitate the functions of an organ and mimic the environment within the body more accurately than current culture systems.
“Whenever cells are grown on plastic [lab] plates, we are creating a static environment for them to grow in. But if you think about cells and tissues within the body, they are always going to experience blood or airflow. It is never a stagnant situation within the body. Those micro channels replicate the fluid flow conditions within the body”, says Dr Jain.
Development of the gut-on-chip technology
The revolutionary organ-on-chip technology was first developed at Harvard and the lung was the first organ to be placed on the chip. Researchers were able to build a chip that captured pulmonary oedema caused by drug toxicity.
It was not long before scientists realised that this same technology could model other organs as well. Today, researchers at the Wyss Institute for Biologically Inspired Engineering at Harvard have modelled over 15 different organs, with the vagina being the latest addition to its repertoire.
The first gut on a chip was developed to imitate the peristaltic movement of the cells in the gut, the same movement that is responsible for pushing food through the intestinal tract during digestion and then later to aid in bowel movements.
There has been huge interest in studying the interactions between the gut and the microbiome. But growing both human cells and microbes together in the same culture system is difficult.
“It has been extremely challenging to co-cultivate human cells with microorganisms as most gut bacteria live without or little oxygen,” says Dr Shin. She further adds that with the gut-on-chip model they can now manipulate the level of oxygen supplied, which enables the growth of anaerobic bacteria along with human cells.
Application of gut-on-chip technology
The development of gut-on-chip technology has made it possible to build models that are otherwise impossible to recreate in a lab setting.
Research is afoot on the role the gut plays in immune response by studying the interaction of gut bacteria with the intestine.
It is also giving researchers a proof of principle to show that the gut-on-a-chip device can be used to create human disease models and gain new insights into gut inflammation.
Not only is the gut being modelled on the chip, but researchers are also connecting multiple organs on the chip to form the gut-brain axis and study how the gut microbiome could affect the behaviour and cognition abilities of the brain.
What the road ahead looks like
The gut-on-a-chip model is being used to better understand human-microbiome interactions, model malnutrition disorders and inflammatory diseases of the gut, and perform personalised drug testing.
However, even though this technology holds a lot of promise to be used as a validated pre-clinical test, it might not be able to completely replace animal models.
“It is already effectively used in some known government agencies and pharmaceutical companies, although the data generated from the organ-on-a-chip platforms do not stand alone yet. It is hard to predict how long it will take to stand alone and replace the animal models” says Dr Shin.
“I believe more efforts by biomedical scientists and more data need to be pooled to try reducing the gap between animal experiments, human clinical trials, and organ-on-a-chip experiments,” she adds.
Another problem lies in getting experts from different disciplines of biology and engineering to come together to make these chips a reality.
“It is still thought that this technology needs special expertise to use in a scientific laboratory. There are more and more efforts to make the platform automated, high-throughput, and user-friendly,” says Dr Shin. She adds that she still feels it will be a good alternative to animal experiments in the future along with becoming more cost and labour effective.
“These technologies need more robust regulatory guidelines for them to be used as a validated preclinical test,” says Dr Jain. Once this is done and more people are trained to use these technologies, it will become easier to adapt them for toxicology studies, she adds.
















One Response
Good article